When reviewing therapeutic classes by cost, rheumatoid arthritis (RA) will likely sit at the top of pharmacy benefit spend for a plan sponsor. The inflammatory condition is also one of the leading causes of disability since RA patients are seven times more likely to reduce work hours. They are also six times more likely to incur medical charges, so the purpose of this article is to help employers gain a better grasp of the condition and insight into current approaches available to efficiently spend health care dollars.
Rheumatoid Arthritis Background
Impacting 1.3 million Americans, RA is a chronic autoimmune disease characterized by painful swelling mainly seen in the joints of the hands and feet. This swelling and pain occurs when the immune system attacks the tissue lining the joints, known as the synovium, that eventually leads to erosion of the joint. The exact cause of the condition is still unknown, but a variety of genetic and environmental factors have been shown to increase the risk of RA, such as smoking and obesity. It also impacts women two-to-three times more than men and the likelihood of diagnosis increases with age. Interestingly, the only risk factor shown to decrease the risk of developing RA is breastfeeding.
Symptoms of RA generally worsen in frequency and severity due to the progressive nature of the disease and can lead to complications in major organs, such as the heart, lungs and kidneys, and in the worst cases, death. If unproperly treated, RA patients may have an average life expectancy shortened by three-to-seven years, while those with persistent high disease activity (severe RA) may pass away 10-15 years earlier than expected.
The active phase of RA can often be sporadic and debilitating and is often referred to as a flare. Many RA patients
experience flares with varying frequency, which is why patients often rotate among the various treatments. A 2013 survey of 1,000 RA patients showed that roughly three-fourths average at least one flare per month with only 4% stating the frequency was less than once a year.
This same survey asked the length of the last experienced flare with 65% reporting the flare lasting at least one week.
Rheumatoid Arthritis Treatment
While there is no cure for RA, the goal of therapy is to reduce joint inflammation and improve the capability to perform daily activities by fostering remission. Many patients will start with non-steroidal antiinflammatory drugs (NSAIDs), such as ibuprofen, and need to change medications and/or add additional therapies to help reduce swelling and slow the progression of the disease. Disease-modifying antirheumatic drugs (DMARDs) are the first-line treatment for RA with the most commonly prescribed being methotrexate. As patients progress through the latest American College of Rheumatology (ACR) treatment recommendations, multiple non-specialty DMARDs are often beset by specialty biologic DMARDs, such as Humira, if moderate to high disease activity persists. If remission occurs, dosage reductions are suggested to minimize the onset of tolerance to the prescribed regimen. This is in line with the ACR recommended dosing in RA approach to “treat-to-target” based on mutual patient-prescriber consideration of patient priorities and given for at least three months before dose increases or decreases to achieve optimal therapeutic response.
Corticosteroids, such as prednisone, may be used to treat unrelenting disease activity as well as flares, but the ACR states corticosteroids should be used at the lowest possible dose for a short duration to offer a favorable risk/benefit ratio. Prolonged use of corticosteroids is associated with a higher mortality risk due to the variety of potential serious side effects, including (but not limited to): osteoporosis, cardiovascular disease, hyperglycemia and immunosuppression.
Impact to Employers
When compared to the population without RA, Americans with the condition have triple the direct health care costs, double the hospitalization rates and ten times the work disability rates. Of the direct costs, roughly two-thirds come from prescription products with the majority of spend occurring on the pharmacy benefit due to the high cost of self-injectable biologics. Studies have shown that the indirect costs associated with RA are slightly higher than the direct costs due to the reduced ability to perform activities at home and work (presenteeism) as well as missing work altogether (absenteeism). About 25% of employees with RA become work-disabled within the first two-to-three years of diagnosis.
Employees with RA face many challenges depending on their symptoms and job responsibilities that their employer could potentially help alleviate via workplace accommodations. A flexible work schedule can aid patients in dealing with morning joint stiffness issues as well as the ability to make appointments with their specialists. It is also beneficial to encourage employees to seek care from an occupational therapist in order to develop strategies to optimize productivity. Since lack of physical activity and smoking negatively impact RA, offering wellness programs and policies to promote healthier living could help slow disease progression.
Strategies to Promote Optimal RA Spend
A comprehensive combination of formulary development, benefit design and utilization management are necessary to control RA spend and trend. Due to RA’s high ranking in plan cost reports, some of the strategies to manage the class may be familiar from previous discussions with vendors, nonetheless it will be beneficial to highlight the range of options.
- Considering over a dozen biological products are available for the treatment of RA, Pharmacy Benefit Managers (PBMs) should be able to leverage competition in negotiations for rebates with manufacturers to reduce the overall net cost to the payer. This is generally done by placing drugs with higher net costs in non-preferred copay tiers or excluding them altogether from the formulary.
- Once the formulary is set, prior authorizations (PAs) with step therapy should be in place to ensure prescribing to guidelines and promoting lower cost alternatives. For example, one program for RA comes from the recommendation in the guidelines from the ACR that three oral generics (methotrexate, sulfasalazine and hydroxychloroquine) can delay the need for a biologic product, such as Humira, by 6-12 months. That recommendation is based on the clinical trial data from a study run by rheumatologists, where the average savings per eligible patient was around $7,500.
- Another way to control spend for RA biologics can be through the utilization of biosimilars, which tend be considered the “generic” versions of brand name biologics. For an in-depth look at biosimilars and strategies to promote usage, visit our website for the “Biosimilar Basics for Plan Sponsors” article of Winter 2016. Since that article was written, multiple biosimilars are now available for intravenous medication, Remicade (infliximab), to help lower costs under the medical benefit where it is typically billed. Unfortunately, infliximab biosimilars have had a slower uptake due to contracting for medical rebates, which are unlikely to be shared with the payer, and physician reimbursement being tied to the average sales price (ASP). As for the pharmacy benefit, we should see about seven biosimilars launched in 2023 for the top selling drug, Humira, with an outside chance one is launched in 2020. The number two selling drug, Enbrel, is in a patent dispute that should be settled by the courts in 2019. The ruling will determine if the biosimilar, Erelzi™, can be launched in 2019 or 2029, so many payers are anxiously awaiting the court’s decision.
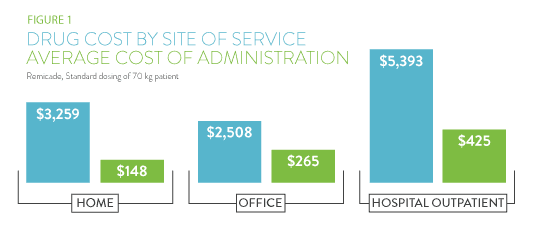
- Since both the medical and pharmacy benefit are impacted by RA drugs, site of care (SOC) alignment to promote the most cost-effective dispensing channel is an often-discussed strategy that has seen little adoption. The first priority is to ensure all self-administered drugs are being dispensed under the pharmacy benefit. For infused products, the three SOCs of outpatient hospital, physician’s office and patient’s home can offer wide varieties in both drug price and cost of administration. A 2013 study by CVS Health of the varying cost of Remicade and its administration can be seen in Figure 1. These results are not uncommon for other infused drugs at the hospital, but the medical policies to steer patients to lower cost sites has been slow. Medical carriers could create a network of Remicade biosimilar providers to utilize when new patients begin RA therapy.
- Value-based contracting is beginning to take hold via PBM and manufacturer negotiations to demonstrate value for existing RA products under the pharmacy benefit. If a drug is unable to control the progression of the
disease or causes nonadherence due to side effects, the PBM should be able to receive additional manufacturer dollars in order to meet therapeutic class trend guarantees for the payer. The inclusion of medical claim data would enhance these types of programs significantly, but it has provided many obstacles for administration. - Finally, specialty pharmacy programs that support patients adhering to these expensive medications are critical in
controlling spend on the pharmacy benefit while reducing long-term implications that could impact the medical benefit.
Similar to how the condition of RA progresses, so have the products and strategies to promote more optimal spend. Hopefully employers and their vendors can utilize these opportunities to support the workforce impacted to enhance the quality of life and productivity of employees.
TO DISCUSS FURTHER, PLEASE CONTACT MATT HARMAN AT [email protected].
Download Article


