Nowadays, it’s nearly impossible to sit down and enjoy your favorite show without seeing at least one pharmaceutical commercial. Of course, you’ll likely see ads for GLP-1s targeting weight loss, but you’re also likely to see another promoting treatment for inflammatory conditions, like psoriasis. With big, manufactured smiles and optimistic testimonials, these commercials make it seem as though these biologic drugs offer quick, life-changing solutions. While they may highlight real success stories, they rarely address the financial complexities that come with these medications.
Four inflammatory conditions alone — psoriasis, psoriatic arthritis, ulcerative colitis (UC) and Crohn’s disease (CD) — account for $27 in total gross cost per member per month (PMPM) across the Employers Health book of business. While newer biologic medications are highly efficacious, they tend to be significantly more expensive than traditional treatments. As the prevalence of these conditions continues to rise and risk factors become more prominent, Employers Health aims to identify a balanced approach to managing these disease states, ensuring access to high-quality therapies for members while containing costs for the plan sponsor.
Inflammation explained
To understand these chronic conditions and their treatment options, we need to understand inflammation. Inflammation is the body’s natural immune response to illness, injury or foreign substances. In a normal immune response, white blood cells are sent to the site of injury to surround and protect the area, causing redness or swelling. For most, acute inflammation lasts only a few hours or days and is usually the result of something minor like scraping a knee or rolling an ankle. In those with autoimmune inflammatory conditions, the body’s inflammatory response is chronic, persisting for months or years, because it mistakenly sends inflammatory cells despite the absence of illness or injury.
In the U.S., nearly 125 million people are living with a chronic inflammatory condition.1 It is predicted the prevalence will only continue to rise with the increase in those experiencing risk factors associated with chronic inflammation, including obesity, exposure to environmental factors, increase in stress and sleep disorders. While there are various types of autoimmune and inflammatory conditions, this article focuses on only a few of those conditions — including psoriasis, psoriatic arthritis, CD and UC.
Skin conditions: understanding the complexities of psoriasis and psoriatic arthritis
Psoriasis, also known as plaque psoriasis for its distinct scale-like patches, is an autoimmune, inflammatory condition that affects 3% of adults aged 20 and older. Typical symptoms include itchy, cracked, dry or discolored skin and those aforementioned scaly plaques. Although there is no cure for psoriasis, there are effective treatment options on the market.2
Treatment largely depends on the severity of the condition and a patient’s comorbidities. Mild-to-moderate psoriasis may be well controlled with topical therapies — over the counter or prescription topical corticosteroids, topical calcineurin inhibitors, vitamin D analogs, etc. — or with phototherapy. Patients with more moderate-to-severe psoriasis may use a combination of topical treatments and a systemic agent. Systemic options include non-biologic therapies that target the entire immune system — such as methotrexate, cyclosporin, acitretin — or biologic therapies that target specific parts of the immune system. The American Academy of Dermatology Association generally recommends biologics to treat moderate-to-severe psoriasis for more targeted symptom control and to reduce risk of broader immune suppression and complications.3
Inflammatory conditions can affect far more than an individual’s skin. Roughly 1 in 3 people with psoriasis may also develop psoriatic arthritis. An estimated 2.4 million Americans are diagnosed with psoriatic arthritis, with more than 15% of people with psoriasis likely having undiagnosed psoriatic arthritis. Psoriatic arthritis can resemble other forms of arthritis, causing joint pain, swelling, tenderness and stiffness. Its severity depends on the number of joints affected and is unrelated to the severity of a person’s psoriasis.
Treatment options are primarily systemic in nature. When developing a systemic drug for psoriasis, manufacturers oftentimes choose to have the drug undergo clinical trials for the additional indication to treat psoriatic arthritis and vice versa. Crossover does exist between the biologic and systemic treatment options available for psoriasis and psoriatic arthritis, as shown in TABLE 1.
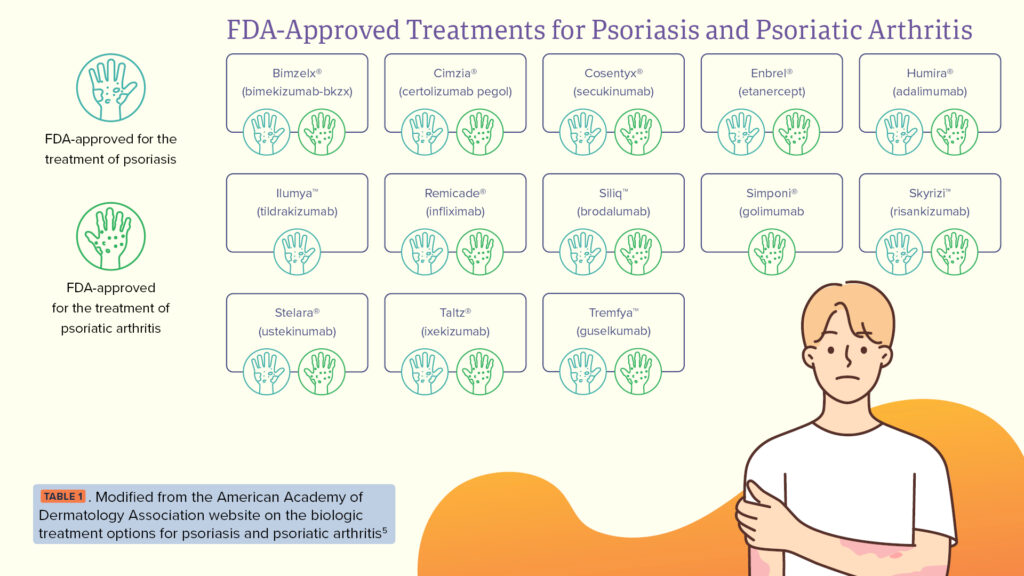
These conditions can significantly impact an employee’s ability to work, potentially leading to absenteeism, reduced productivity or job loss. In the U.S., psoriasis and psoriatic arthritis contribute to an estimated $35 billion in health care costs annually, with $11.2 billion stemming from productivity losses (32%) and $11.8 billion from reduced health-related quality of life (34%).4 Across the Employers Health book of business for 2024, these conditions accounted for approximately $15 in total gross cost PMPM, with those costs rising each quarter. This trend is heavily driven by drug price inflation, new utilizers and increased utilization of higher-cost biologic products including Skyrizi® and Tremfya™.
Inflammation in the gut: a closer look at ulcerative colitis and Crohn’s disease
Another type of inflammation, often not visible externally, affects the gastrointestinal (GI) tract and is classified as inflammatory bowel disease (IBD). IBD is an umbrella term for GI conditions, including two of the most common IBDs, UC and CD. The two conditions are chronic inflammatory diseases of the GI tract but differ in the specific area where inflammation occurs. In CD, inflammation can involve all layers of the bowel wall and occurs anywhere in the GI tract, from the mouth to the anus. In UC, inflammation is limited to the inner lining of the colon. In both conditions, patients may experience gut-related symptoms, such as abdominal pain, changes in their stool frequency, blood in their stool, weight loss, fatigue or vomiting. While patients can be diagnosed with IBD at any age, most patients are diagnosed between ages 15 and 35.
An estimated 2.39 million Americans have IBD, with roughly 1.25 million diagnosed with UC and 1.01 million with CD.6 Treatment for moderate-to-severe cases has rapidly evolved in the past few decades. With the evolution of treatment options comes a rise in the cost of care for IBD. In 2018, the overall costs for IBD-related care were $8.5 billion with 71% of those costs stemming from prescription medications. A recent study published in Clinical Gastroenterology and Hepatology estimated overall IBD care costs in high-income countries range from $9,000 to $12,000 per person annually and depend heavily on patient location and disease severity, with higher costs experienced by moderate-to-severe patients in the U.S. This estimate is solely based on direct costs of IBD and does not account for costs associated with overall quality of life or indirect health care costs. The study notes that historically, costs were caused primarily by inpatient care, surgeries or hospitalizations; however, in recent decades a majority of costs have shifted to outpatient care and newer biologic therapies.
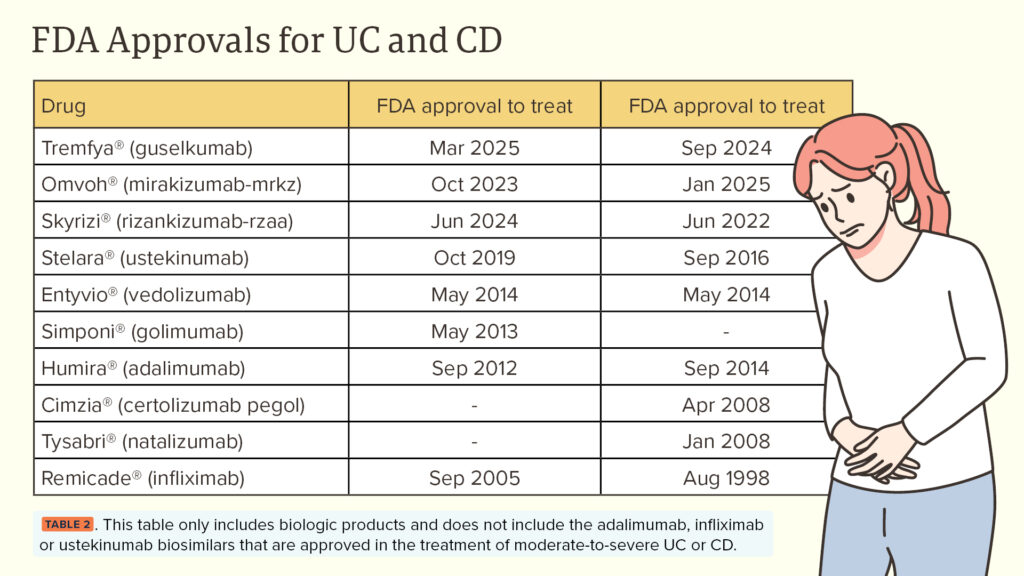
The first biologic therapy approved for the treatment of moderate-to-severe IBD was Remicade® (infliximab), receiving approval for CD in 1998, and UC in 2005. TABLE 2 showcases recent FDA approvals in these disease states and how the landscape of IBD is evolving. As these newer therapies hit the market, the current American Gastroenterological Association (AGA) guidelines recommend a biologic-first approach to treatment, considering patient-specific factors and other trialed therapies.
Costs and considerations
As the prevalence of these inflammatory conditions rises, there is a continued focus on the management and containment of costs for treating these diseases. Despite biosimilars already having entered the market, in February 2024, Humira® retained 96% of the market share. It wasn’t until April 2024, when CVS became the first pharmacy benefit manager (PBM) to exclude Humira® from its template formularies in favor of the biosimilar, Hyrimoz®, that the market began to shift. By the end of 2024, Humira® accounted for only 72% of the adalimumab market with biosimilars accounting for 23% of the market share.7 Across the Employers Health and CVS shared book of business, there was over a 90% conversion rate to Humira® biosimilars, which offer an 80-83% list price discount per prescription compared to Humira®. Optum Rx and Express Scripts followed suit, excluding Humira® from all formularies effective January 2025.
Holding roughly 55% of the market share in the Employers Health book of business for the treatment of UC and CD, Stelara® biosimilars represent the next big opportunity for savings. Wezlana® was the first Stelara biosimilar launched in January 2025. All Stelara biosimilars that have launched so far claim to offer a significant discount of 80-90% off the list price. The big three PBM’s Stelara® biosimilar decisions are mirroring those of the early Humira® biosimilar decisions, first placing the biosimilars at parity and then moving to exclude the reference product.
Optum Rx was first to place the biosimilar Wezlana® (by Nuvaila) on its template formularies at parity with Stelara® on January 1, 2025. Similarly, Express Scripts announced its plans to add ustekinumab-ttwe (by Quallent), Yesintek® and Selarsdi® at parity with Stelara® throughout the first half of 2025. Although first to market with its Humira® biosimilar decision, CVS was the last of the big three to announce its Stelara® biosimilar strategy, placing Pyzchiva® (by Cordavis and Sandoz) and Yesintek® at parity with Stelara®, effective July 1, 2025. Out of these three, Optum Rx will be the first to exclude Stelara® with preference toward the biosimilars also effective July 1, 2025. Since a majority of the Stelara® biosimilars are coming to market with interchangeability status and FDA approval for the same indications as the reference product, transitioning patients from Stelara® to a biosimilar should be less complex and less nuanced than the shift to Humira® biosimilars.
Looking ahead
As we start to see the movement of the Stelara biosimilar products entering the market, Employers Health’s clinical team is seeking additional avenues to encourage biosimilar adoption and early opportunities for savings. This includes exploring a biosimilar first strategy for the aforementioned conditions to promote early biosimilar treatment before moving to higher-cost biologic therapy options based on current clinical treatment guidelines. For additional insight, register for the Employers Health Inflammatory Insights webinar presented at the 2025 Annual Benefits Forum by clinical advisors Catherine Berger and Ernesto Munoz here.
